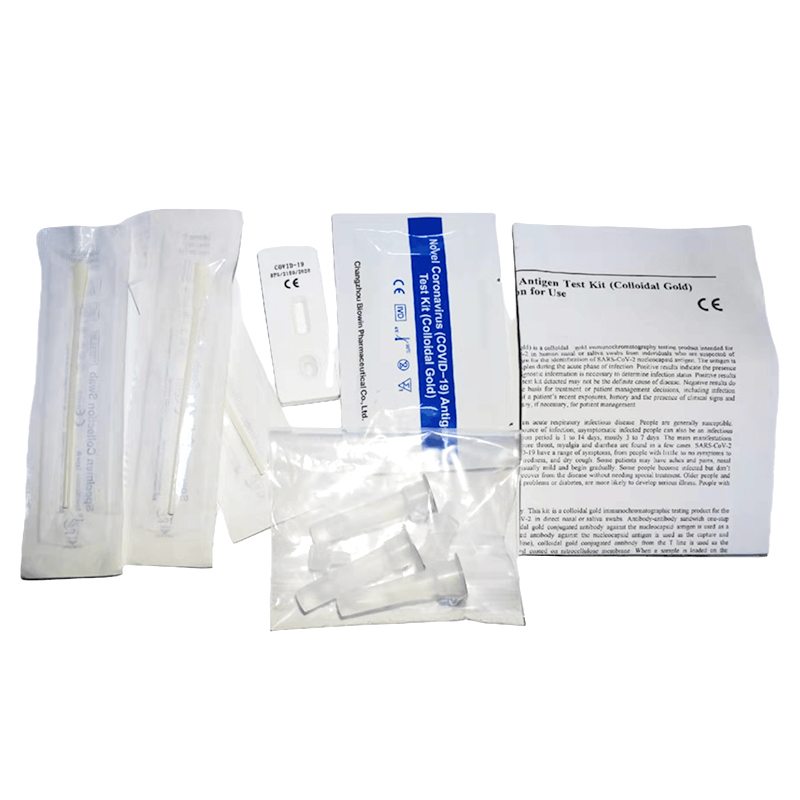
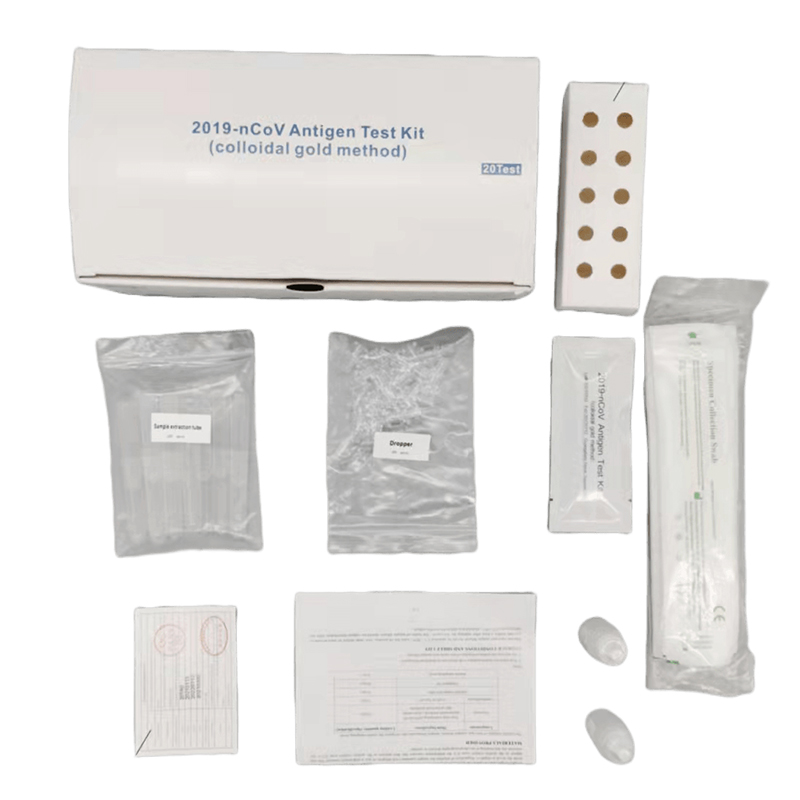
The monkeypox virus antigen detection kit is used to detect monkeypox virus in serum or pathological exudate samples.
-PCR fluorescence probe method.
This product is based on real-time fluorescent PCR technology. The highly protected region of the partial coding region of the monkeypox virus gene is selected as the target region, and specific primers and fluorescent probes are designed for PCR amplification to qualitatively detect the monkeypox virus DNA in the patient’s serum and the lesion extract (vesicular fluid, pustular fluid). It is applicable to the auxiliary diagnosis of related diseases caused by monkeypox virus infection.
{Product features}
The reagent is equipped with an external internal standard detection system to detect the nucleic acid extraction and PCR amplification process and reduce the occurrence of false negative results.
The sample can be extracted to the result report within 70 minutes.
The sensitivity of the product reaches 200 compies/ml.
High specificity and repeatability, wide detection range.
It has obtained EU CE certification.
{usage method}
Serum sample collection: serum should be separated as soon as possible after blood collection to avoid hemolysis. The samples shall be tested immediately after collection. If it cannot be detected immediately, the serum sample can be stable for 5 days when stored at 2-8 ℃; The sample can be kept at – 20 ℃ or lower temperature for a longer time, and repeated freezing and thawing are avoided.
Collection of pathological exudate samples: use the sample collection swab to take pus and secretions from the lesions and send them to the extraction tube. The specimen should be tested immediately. If it cannot be tested immediately, it should be frozen at – 20 ° C to – 80 ° C.
{Product parameters}
Test principle: fluorescent PCR
Sample type: serum, lesion extract (vesicular fluid, pustular fluid)
Storage condition: keep away from light – 20 ± 5 ℃
Valid period: good stability, valid period not less than 12 months
Type and specification: 24 persons/box; 48 persons/box; 96 persons/box